Polycythemia vera (PV)
What is polycythemia vera (PV)?
Polycythemia vera (PV) is a chronic, progressive myeloproliferative neoplasm (MPN) primarily characterized by an elevation of the red blood cells. The disease is most common in men over the age of 60, but anyone can develop PV. PV patients typically experience an elevated leukocyte (white blood cell) count, an elevated platelet count and an enlarged spleen, especially over time.
Watch an overview video of the diagnosis, presenting symptoms, disease course, and treatment options of polycythemia vera for patients, their caregivers, and their loved ones created by Dr. Ruben A. Mesa and Dr. Robyn M. Scherber of UT Health San Antonio, MD Anderson Cancer Center.
View Nutrition Recommendations for MPN Patients, created by Dr. Robyn Scherber, MD; Dr. Ruben Mesa, MD; Ryan Eckert, MS, Mays Cancer Center at UT Health San Antonio MD Anderson MPN Quality of Life Study Group.
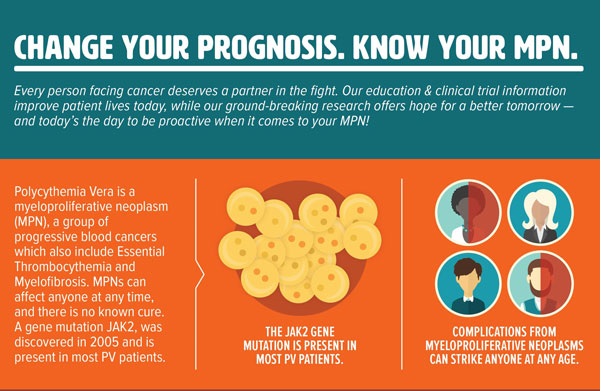
What causes PV?
The trigger for polycythemia vera (PV) and other myeloproliferative neoplasms (MPNs) isn’t known. However, researchers have discovered that PV and other MPNs may be caused by non-inherited genetic mutations affecting proteins that work in signaling pathways in cells.
Nearly all PV patients have a mutation called “JAK2V617F” (found in the JAK2 gene) in their blood-forming cells. This mutation is one of the ways that JAK (Janus kinase) pathway signaling can become dysregulated and cause the body to produce too many blood cells.
Risk factors associated with PV include:
Sex
Men are slightly more likely than women to develop the condition.
Age
People older than 60 are most likely to develop the condition, though it may occur at any age.
Environment
Exposure to intense radiation may increase the risk for the condition. It has also been suggested that exposure to toxic substances may increase risk for PV.
JAK2 mutation
Approximately 95% of all PV patients have a mutation of the JAK2 gene in their blood-forming cells. This mutation leads to hyperactive JAK (Janus kinase) signaling, causing the body to make the wrong number of blood cells.
How is PV diagnosed?
For many patients, PV is initially diagnosed after a routine complete blood count (CBC) reveals in increase in the red blood cells, sometimes in conjunction with an increase in white blood cells and/or platelets.
In other instances, the initial diagnosis occurs when a physician identifies signs of the disease (e.g., redness to the complexion, blood clotting or an increase in the size of the spleen) during a routine medical examination.
Common diagnostic tests for polycythemia vera include:
Blood tests
A complete blood count can identify an increase in hemoglobin as well as white blood cells and/or platelets.
Bone marrow biopsy
A bone marrow biopsy may reveal an excess of precursors to red blood platelets, red blood cells and white blood cells.
Gene mutation analysis of blood cells
Approximately 95% of PV patients test positive for a JAK2 gene mutation. An analysis of red blood cells may also indicate a low erythropoietin level (a hormone involved in red blood cell production).
Low erythropoietin level
An analysis of red blood cells may also indicate a low erythropoietin level (a hormone involved in red blood cell production).
What are the symptoms of PV?
Like patients with other types of MPNs, PV patients may present with a wide range of symptoms or exhibit no symptoms at all.
Common symptoms of PV include:
- Headache
- Sweating
- Ringing in the ears
- Blurred vision or blind spots
- Dizziness or vertigo
- Reddish or purplish skin
- Unexpected weight loss
- Bleeding or clotting
- Early feeling of fullness (satiety)
- Itching (pruritis), especially after taking a shower
- Burning and redness of the hands or feet
- Tiredness (fatigue)
- Night sweats
- Bone pain
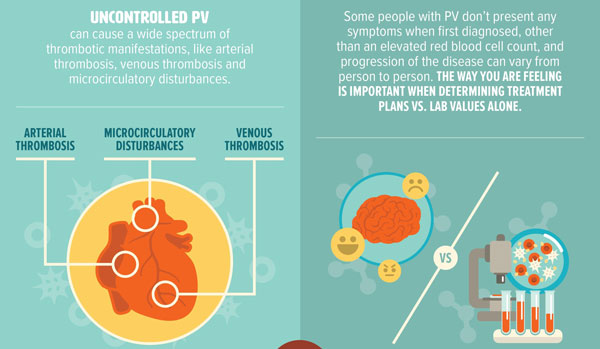
What is the prognosis for PV?
Most PV patients enjoy longevity, if they receive regular monitoring and treatment. However, in some cases, prolonged survival can be challenged by the development of other syndromes.
Approximately 15% of PV patients develop myelofibrosis, a progressive bone marrow disorder that results in bone marrow scarring, severe anemia, and enlargement of the liver and spleen. This change can be heralded by the onset of anemia, or a low red blood count, as opposed to high red blood counts, and a significant increase in the size of the spleen). In a smaller number of cases, PV may progress to acute leukemia (AML).
Additional complications that can occur with PV include arterial thromboses (heart attacks, strokes, intestinal gangrene), venous thromboses (of the portal and/or hepatic veins), or pulmonary embolism.
Patients at risk of developing these illnesses should have their blood counts routinely monitored and controlled by a physician.
What are the available treatments for PV?
Many of the treatment options for PV are designed to manage the disease by returning hematocrit levels to normal values. However, the circumstances are different for every PV patient and asymptomatic patients may not require treatment for extended periods of time. Routine monitoring by a physician is recommended for all PV patients, regardless of symptoms.
When treatment is required, common treatment options for PV include:
Phlebotomy
Phlebotomy is the removal of blood to reduce the number of blood cells. With fewer blood cells, the blood is thinner and flows more easily, improving symptoms and reducing the risk for blood clotting. This procedure is typically done to meet target blood count goals that are determined by the physician, taking into consideration the patient’s sex and other factors.
Low-Dose Aspirin
Most, if not all PV sufferers are prescribed a low-dose aspirin treatment. Since aspirin prevents platelets from sticking together, it reduces the occurrence of blood clots that can cause life-threatening heart attacks or strokes.
Combined with low-dose aspirin, the regular maintenance of a hematocrit below .45 for men and .42 for women is currently accepted as a non-leukomegenic approach and a first choice treatment for recently diagnosed, low-risk PV patients.
If phlebotomy and low-dose aspirin are not effective or appropriate, or if a patient is consider higher risk for blood clotting, physicians may prescribe medicine to lower red blood count and relieve symptoms, including:
Hydroxyurea
Hydroxyurea is often prescribed for PV patients at high risk for blood clots, based on age and prior history of blood clotting.
Jakafi (ruxolitinib)
Jakafi is the first FDA-approved treatment for PV patients who have an inadequate response to or cannot tolerate hydroxyurea. Jakafi inhibits the JAK 1 and 2 enzymes that are involved in regulating blood and immunological functioning. It also helps decrease the occurrence of an enlarged spleen (splenomegaly) and the need for phlebotomy. Patients do not need to be JAK2 positive to take Jakafi, though the great majority with PV harbor this mutation.
IncyteCares can help people with or without insurance coverage understand their coverage (if any), explore financial assistance options, and provide ongoing support. Incyte is the company that developed Jakafi.
Some PV patients have experienced anemia with or without splenomegaly while on Jakafi. Please discuss in depth with your doctor upon making any treatment decisions.
BESREMi (ropeginterferon alfa-2b-njft)
BESREMi was approved by the FDA in November 2021 as the first interferon specifically approved to treat adults with polycythemia vera regardless of their treatment history. BESREMi is a monopegylated, long-acting interferon designed to be administered once every two weeks (or every four weeks with hematological stability for at least one year). The PharmaEssentia SOURCE program is available to support patients prescribed BESREMi. For more information, visit www.pharmaessentiaSOURCE.com. PharmaEssentia is the company that developed BESREMi.
Pegylated Interferon
Younger patients who require treatment and women of childbearing age are often treated with pegylated interferon because it has not been shown to cause birth defects. Since Pegasys was developed for Hepatitis C and not MPN, it is considered an “off-label” medication. There are several clinical trials currently being conducted to evaluate Pegasys in people with MPNs.

It’s time to change your PV prognosis
MPN Research Foundation is committed to providing education and support to PV patients and their families. In the fight against PV, information is power. Subscribe to receive news and updates about the latest PV treatment options and other resources to change your PV prognosis.
Join the MPN Research community

RON
POLYCYTHEMIA VERA (PV)
Learn how Ron continues his cycling passion despite his PV diagnosis.
